Larissa V. Rodriguez, MD, presented “Stem Cell Therapy – Bench to Bedside” at the Jackson Hole Seminars on February 7, 2018 in Jackson Hole, Wyoming.
How to cite: Rodriguez, Larissa V. “Stem Cell Therapy – Bench to Bedside” February 7, 2018. Accessed [date today]. https://grandroundsinurology.com/Stem-Cell-Therapy-Bench-to-Bedside/
Summary:
Larissa Rodriguez, MD, summarizes the current understanding and available options of stem cell therapy in the context of female pelvic medicine and reconstruction, especially for treating stress urinary incontinence (SUI), pelvic organ prolapse, interstitial cystitis, and fecal incontinence.
Stem Cell Therapy – Bench to Bedside – Transcript
Click on slide to expand
Stem Cell Therapy – Bench to Bedside
I’m going to try, in the next 20 minutes, to give you a summary of where we are in terms of stem cell therapy for areas in female pelvic medicine and reconstruction. So, it’s a broad amount of literature I’m going to try to summarize here for you.
GOALS
The goals would be to talk about stem cell therapy for stress urinary incontinence, for pelvic organ prolapse, for interstitial cystitis, and fecal incontinence.
Urinary Incontinence: Are we Making Progress?
The big bulk is going to be for urinary incontinence, which is where we have been working on for the longest amount of time, and we have more data, and more human data as well.
Vaginal Delivery
So at the time of vaginal delivery, there is significant injury to the pudendal nerve, tear or avulsion of the levator musculature, and ischemia that can lead later on to pelvic floor disorders including prolapse and incontinence.
Changes with Aging
In addition, there are changes that occur with aging where there is atrophy of the skeletal rhabdosphincter, atrophy of the smooth musculature of the intrinsic sphincter unit in women, loss of neuronal mass, and with the lack of estrogen after menopause, there is decrease in vascular channels that help close the urethra. There are vascular channels with the smooth muscle that kind of help with the cooptation and the urethra.
Contributing Factors
So, there are many reasons why women leak urine. Anatomic lack of support, skeletal rhabdosphincter accounts for likely 50% of the continence mechanism in women. There are some studies that demonstrate that as a smooth muscle, the intrinsic smooth muscle of the urethra not only gives it tone, but it accounts for continence in about 40% of the continence mechanism. There are structural changes to the pelvic floor that occur with delivery and aging. There are changes in the sympathetic and parasympathetic innervation. There are pudendal nerve injuries that occur at the time of vaginal delivery, vascular injury, and again, there is a central nervous system component with how we coordinate voiding. So, the optimal treatment for stress urinary incontinence would be personalized to the particular injury that a woman is suffering, which is not the way we treat urinary incontinence right now.
TOMUS
If we look just at one of the large clinical trials looking at randomized patients treated with mid urethral sling, in this particular case, the TOMUS trial that looked at TVT to TO. And, the only reason I put this slide here is to let you know that, although we have great treatment choices for the treatment of stress incontinence, they are not perfect. When we looked objectively at this data, the subjective cure rate was between 55% and 62%. There is definitely room for improvement. There are some complications that we are seeing, and this is short term and about a year after surgery.
Increase Urethral Resistance
There is room for improvement. What we do currently with our gold standard of doing a sling for patients with stress incontinence is that we kink and increase the urethral resistance. We provide backboard support, we can bulk with materials, but none of these treatments really improve the function- the ability- of that urethra to close and open, what it needs to do.
Stem Cells to Treat SUI
So, that has led many of us to look at alternative treatments to see can we really improve the urethral function.
Goal of Tissue Engineering for SUI
So, the goals of tissue engineering strategies for the treatment of stress incontinence have focused on replacing the atrophic smooth muscle, or the skeletal muscle, improve innervation, and improve vascularity with overall the idea will be to restore the function of the urethra.
Cell Based Therapies for SUI
To do this we need cells, maybe. We need a delivery system, maybe. We need a good animal model that represents the disease process. We need to show the cells that we are using survive, what do they become, and to see if any of these areas I’m arguing are important, neovascularity and innervation, are really restored. Lastly, can we really restore the function of the urethra?
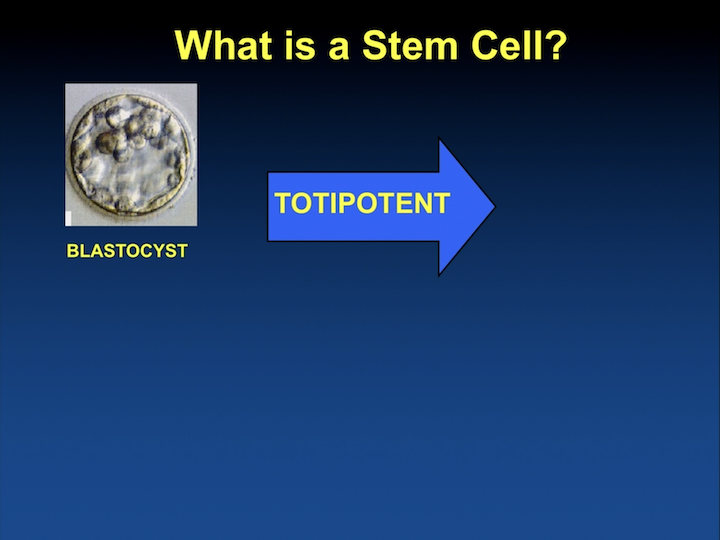
What is a stem cell?
So, when we look at a definition of a stem cell, at the time of fertilization the blastocyst has outside the trophoblast and the inner cell mass, which really provides a totipotent ability to develop an individual. In that case, my youngest son, which is now 14 years old.
What is a stem cell? #2
If we only look at the inner cell mass, it’s what we call embryonic stem cells. You cannot get a whole individual. You don’t have a placenta, but you have cells that are totipotent or multipotent.
What is a stem cell? #3
These cells are immortal. They are identical. They can lead to long-term self renewal, and they can differentiate into all cell types. And the stereotypical cell that can do that is the embryonic stem cell. Maybe some of the induced cells that can be induced to behave as embryonic stem cells. The majority of the research in stress incontinence has not been using these cells. The majority of the research to date has been using adult derived stem cells for a number of reasons.
Adult Stem Cells
There are stem cells in every part of the body, and they are felt to be there to repair, to repair after injury. There are some people looking at stem cells in these particular milieus, these particular regions, looking at cancer. They are felt to also be involved in the development of prostate cancer and other things, but these cells are there for repair.
Advantages of Adult Stem Cells
The advantage of using them is that they can be expanded in culture. There’s no significant ethical or political considerations. Because they are autologous, there is no need for immunosuppression, and this is a big one when we’re dealing with a quality of life benign condition. We don’t want to give a therapy where a woman would have to be immunosuppressed for life when we use a particular embryonic stem cells that doesn’t belong to them. And, they are multipotent, so they can become many tissues.
Stem Cells for SUI
In terms of, to summarize the pre-clinical studies that have been done to date, all of these cell types have been used, umbilical cord stem cells, bone marrow, skeletal muscle. They have a number of different names, skeletal muscle-derived cells, muscle-derived cells, muscle progenitor cells, satellite cells. They are all pretty much the same cells. And adipose cells that have been called also a number of different names, adipose-derived stem cells or adipose stem cells.
Bone Marrow Stem Cells
Bone marrow stem cells have been used in an acute model of urethral injury to give them intravenously to show that these cells can home to the area of injury.
Bone Marrow Stem Cells #2
There’s a couple of groups that have been looking at this kind of intravenous delivery of cells. I question the relevance of this model since the injury happened at the time of delivery, but really, the symptom happens two or three decades later. But, they have also been used for periurethral injection in more chronic models.
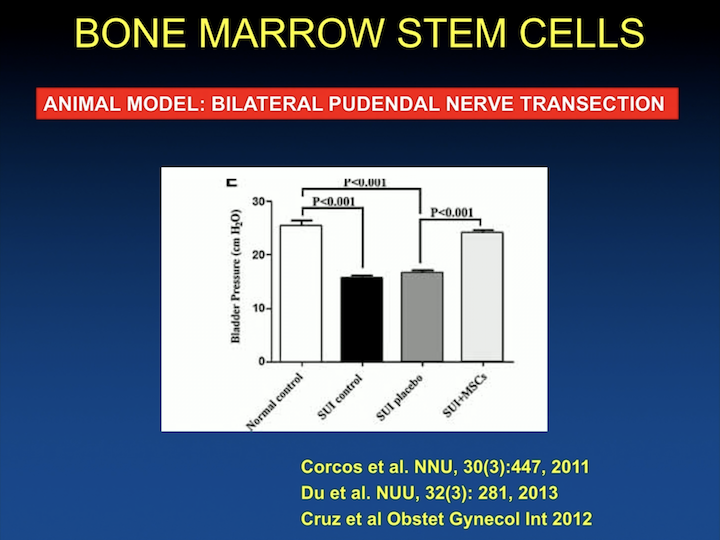
Bone Marrow Stem Cells #3
And, the data as in almost all of the pre-clinical studies show that you can in different animal models, in this case a bilateral pudendal nerve transection rodent model, that when you inject cells, you have increase in leak upon pressure.
Problems with Bone Marrow Stem Cells
The problem with bone marrow stem cells is the morbidity of the harvest, really going to the bone marrow to get them, and you obtain very few cells, and you have to expand them significantly in the laboratory.
Muscle Derived Stem Cells
The majority of research for the treatment of stress incontinence has been in the use of muscle derived stem cell for the idea of repairing of the rhabdosphincter.
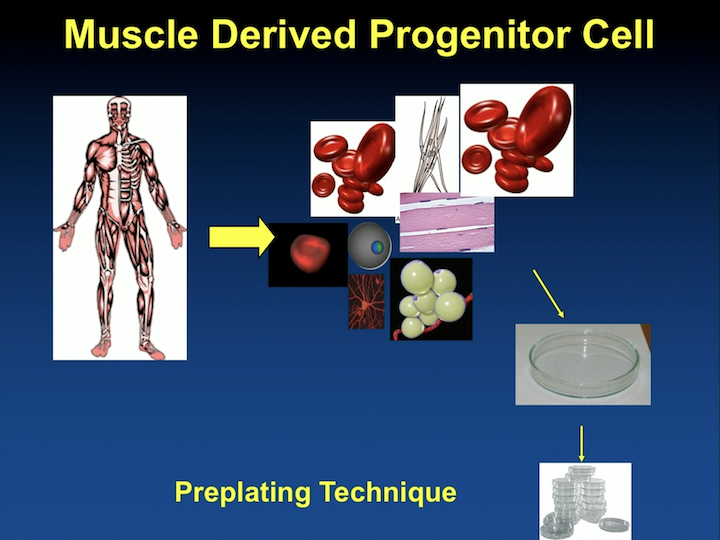
Muscle Derived Stem Cells #2
In this model, a biopsy is obtained either from the thigh or from the bicep, and these cells are then pre-plated. They have to be taken to the laboratory for about 30 to 45 days until you get a more pure population that you can use for research.
MDSC and SUI
There are over 10 studies looking at these cells and stress incontinence in rodents, and there are also studies using non-human primates and canine. There’s a number of different animal models, and it’s hard to compare some of these studies. Some used a urethrolysis by electrocautery model, sciatic nerve transection, and pudendal nerve transection are models of simulated vaginal delivery, and the cells are usually injected in saline or HBSS, which is a saline-like substance, and the longest follow-up has been 6 to 12 weeks.
Muscle Progenitor Cells
Again, as in the previous data you- in the left side corner, you have leak point pressure in controls, and in animals that are injected with cells at different time points. And, you can see that leak point pressure increases in the right side column in each one of these time points, which are the cells being injected. Studies comparing them to fibroblasts show similar results, except fibroblasts cause obstruction.
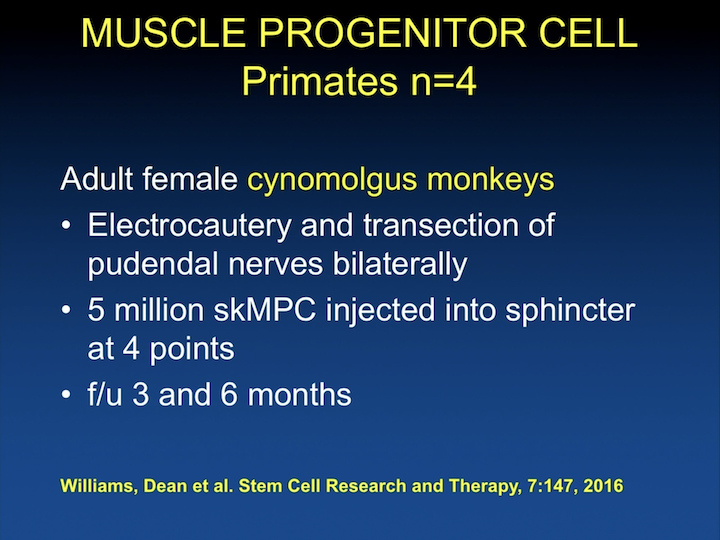
Muscle Progenitor Cell
I think this is really interesting research. It’s pretty recent. Most of this is from 2016 and 2017 with the use of primates. There have been about four publications with the use of primates, and the reason I think it’s interesting, it’s not so much the use of the model or the incontinence of the cells. They’ve been using skeletal muscle derived stem cells and follow these animals at three to six months.
Muscle Progenitor Cell #2
And, they have shown, just like in the rodents, that there are increases in maximal urethral pressure, and that these animals have improvement in their continence at six weeks after local injection. But, they have been able to look at acute versus chronic. So, when they actually do the injury but inject the animal six months later, which is really what we are looking at in humans, the response rate is not as good. So, in an acute setting when you do the injury and you inject the cells, and follow the animals, which is the ability we have in rodents. They see a response, but when they actually wait it out, leave the animal incontinent six months and inject them, the outcomes are not as good. They also have shown, as expected, very minimal response to intravascular delivery of the cells, some when the injury is acute, but again, almost none when the injury is chronic.
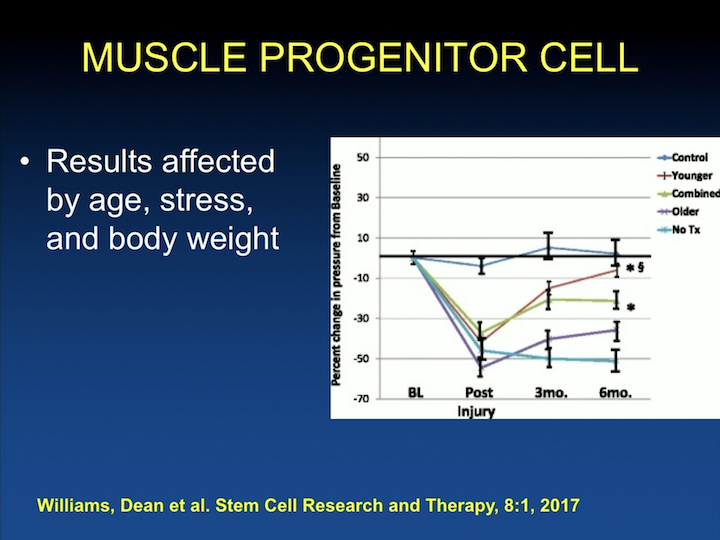
Muscle Progenitor Cells #3
And they have been able to look at other things that I think are going to important when we transfer this to the clinic looking at age, so older animals really don’t do as well. Younger animals respond much better to the therapy with increases in leak point pressure. Stress– so in monkeys, you can have the dominant monkey and all of the submissive monkeys, so the dominant monkeys do better with treatment with stem cells and much better than the other groups. And body weight. The more obese the animal, the less well they do. So, these are things, again, for us to start thinking about as we start translating this to the human population, things that are going to be factors. We talked about replacement of the skeletal muscle in a lot of this research. We have particularly been interested in replacing the atrophic smooth muscle of the intrinsic urethra.
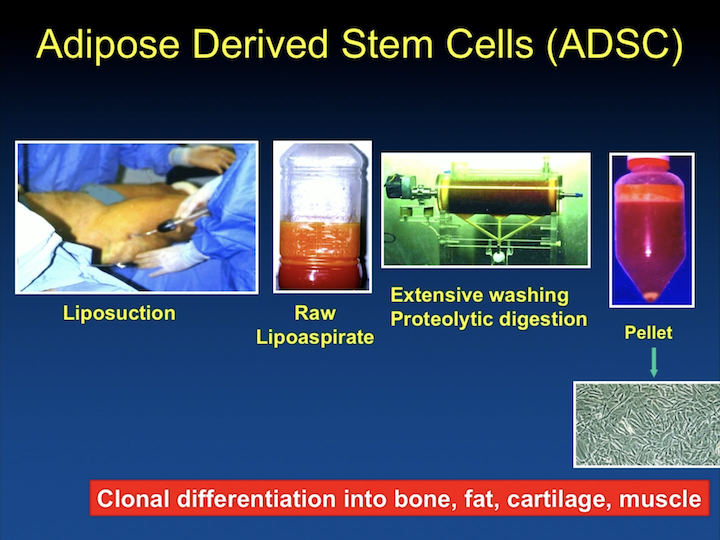
Adipose Derived Stem Cells (ADSC)
And many years ago we started using adipose derived stem cells, and originally in our research we were interested in being able to differentiate these cell into smooth muscle, and make them work like smooth muscle.
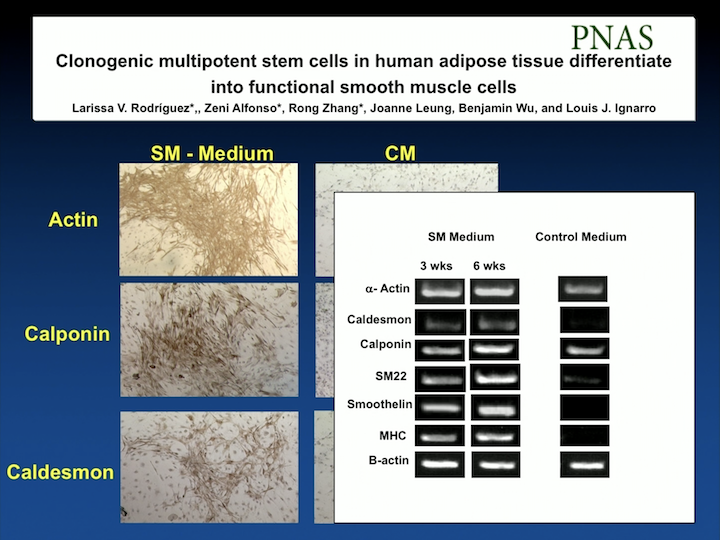
PNAS
We published this work in the Proceedings of the National Academy of Science. We were the first group to be able to differentiate these cells into smooth muscle with markers, and also make sure that the cells actually contracted and relaxed to different pharmacologic agents, as smooth muscle cells do.
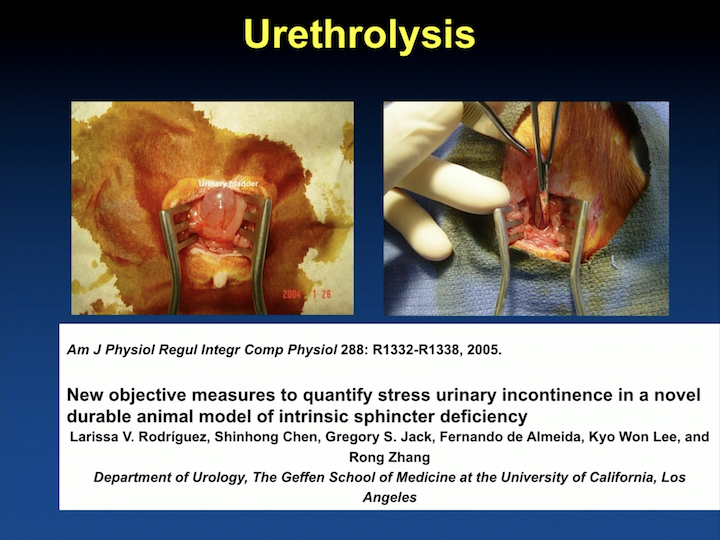
Urethrolysis
We also published one of the first models of incontinence after the well-validated vaginal delivery model that was developed by Tom Lue at UCSF. We looked at urethrolysis, and this is a different. I was at that point interested in a model that was long lasting. I felt that, if we’re going to use stem cells, if we’re going to do any of this type of research, we really have to prove that we’re doing something significant. And, the problem I had with the vaginal delivery model is that it’s a mild model that a lot of the animals recover with time. So, I remember being really young and naïve, and going and spending a couple of days there saying, “I’m never going to convince anybody that with this mild model – – cells they’re going to get better.” I need something more drastic, so we would develop a model to do that and spent a fair amount of time with the idea of differentiating these cells.
ADSC Delivery to Urethra
Delivering it into the animal and making sure they stayed there and behaved like smooth muscle cells.
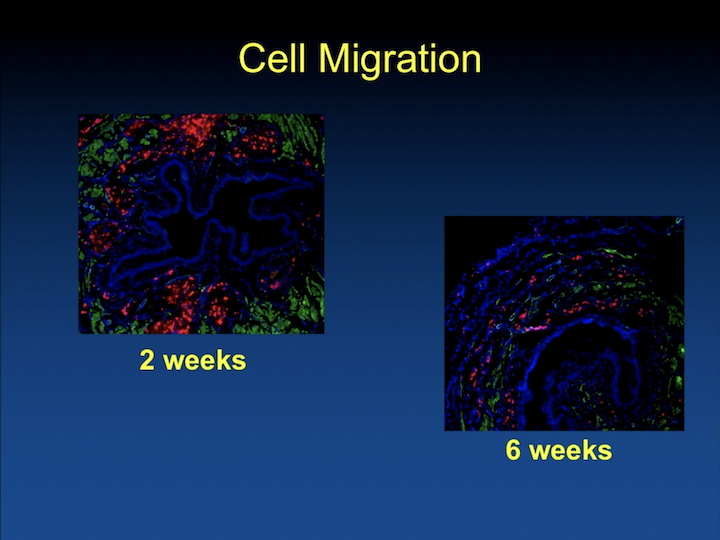
Cell Migration
Well, we quickly found that if we just delivered them in saline the cells are there, and a couple– after some weeks, they are not there anymore. And, I think this is well known at this time, in all types of cell research, in every part of the body.
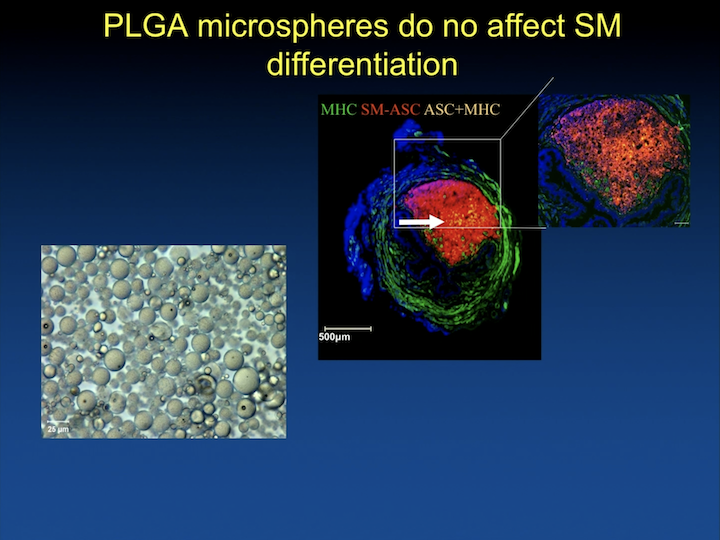
PLGA microspheres do not affect SM differentiation
And we have spent a lot of time working with biomedical engineering, and particularly biomaterials that can help us differentiate the cells in vivo, keep them in place for them to do what they’re supposed to do.
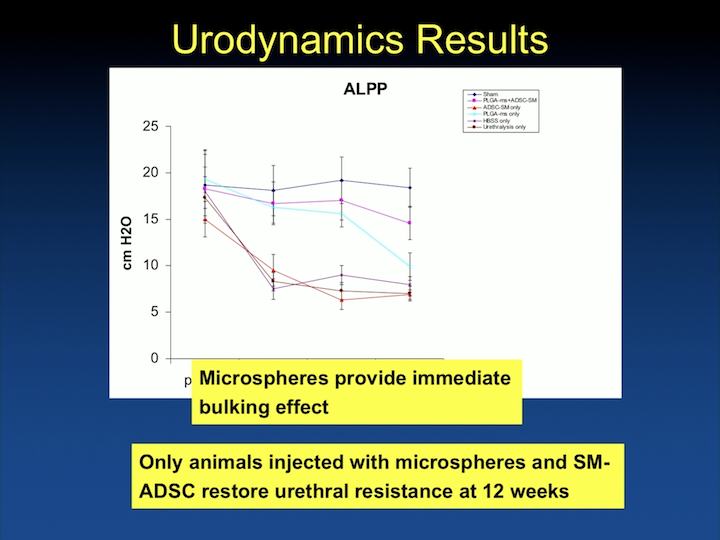
Urodynamics Results
And we are able to show that, in different groups, when we inject animals with biodegradable beads, that these cells do hang around for a long time, and that the beads provide immediate bulking effect.
Urethral Function Carbachol Response
But, that only the cells- the animals injected with smooth muscle differentiated adipose cells- have increase in urethral resistance at 12 weeks, and that this translates in organ bath to actually contractility of the urethra.
ADSC and SUI
There have been a number of different laboratories looking at adipose-derived stem cells. There are, again, a number of different animal models that have been used and different delivery systems.
ADSC and SUI #2
And, all the rodent models show improvements in leak point pressure, and some have shown improvement in urethral function.
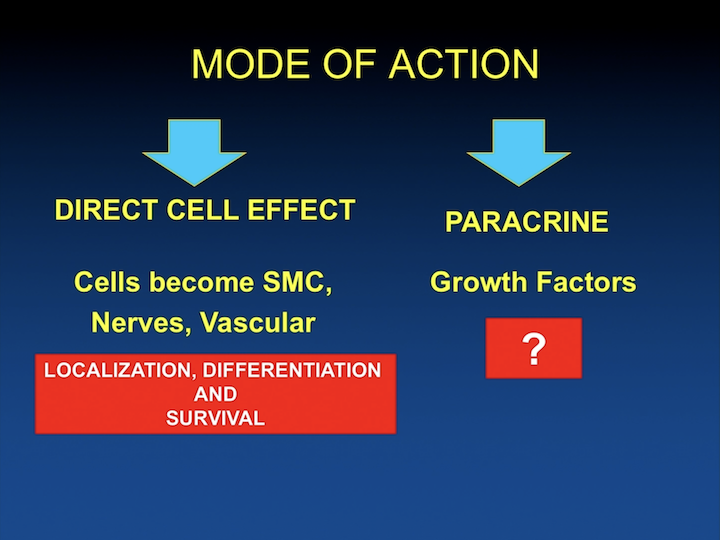
Mode of Action
As time went by, we were interested. Like really, are we going to spend, is a patient going to give a biopsy and you tell them, “Come back in a month and this is going to cost all of this money because you have to expand the cells?” And, I think this is a significant limitation of some of the things that are coming in the pipeline, and we were really working can you do something in the operating room where you harvest the cells, and you put them back in right there and then. So, the patient goes home, and you are done. You don’t have to really expand the cells. So, this whole step where we were differentiating cells into smooth muscle and something else, can we obviate that? It’s also become more apparent that maybe you don’t need the cells at all, that the cells secrete stuff that causes paracrine kind of growth factors that can help localize the local stem cells, but also help with smooth muscle regeneration, skeletal muscle regeneration, and you might not need the cells.
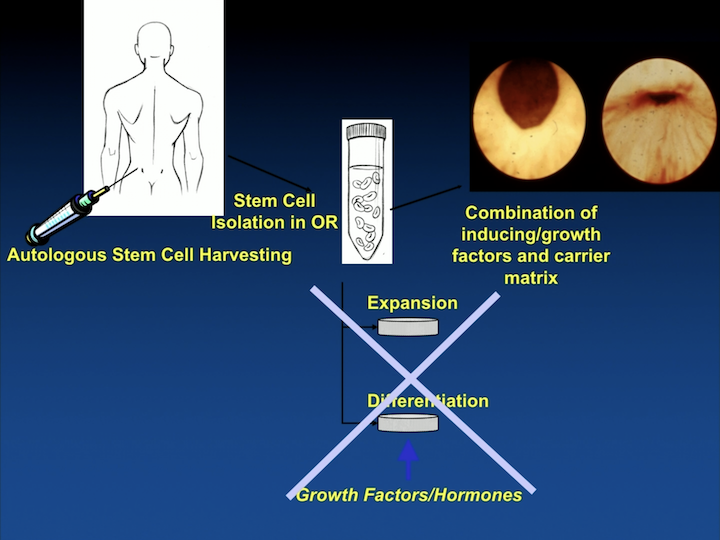
Autologous Stem Cell Harvesting
So a lot of what we are working on is to try to eliminate the expansion outside the body of cells and the differentiation, and maybe start working as to what is it that the cells do, and can we replicate that with injectable materials where we might not need to use the cells. So, a slightly different way than other labs are looking at that, but people are looking at this in many different fashion to really try to come up with a product where we can help our patients.
Human Trials – Austria
There have been a number of human trials. The first one was published in 2007. It was a big deal at the AUA. It was published in Lancet.
Human Trials – Austria #2
It was a study from Austria, and unfortunately, this was retracted due to IRB and other issues with experimental protocol, which set the field back a little bit.
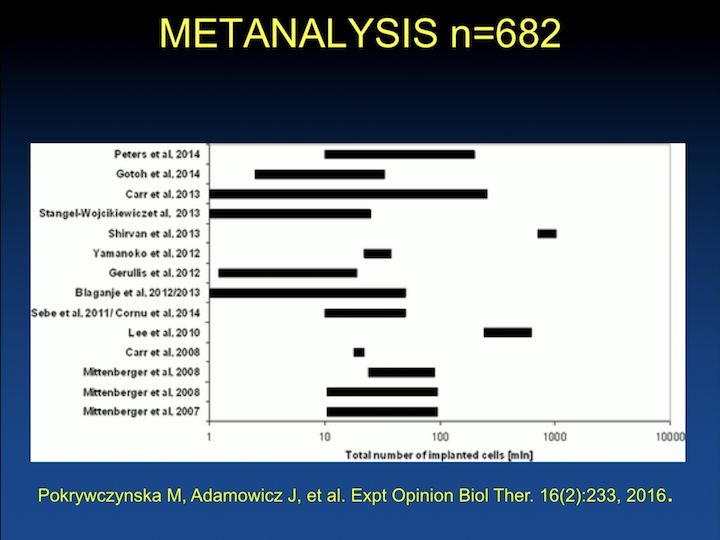
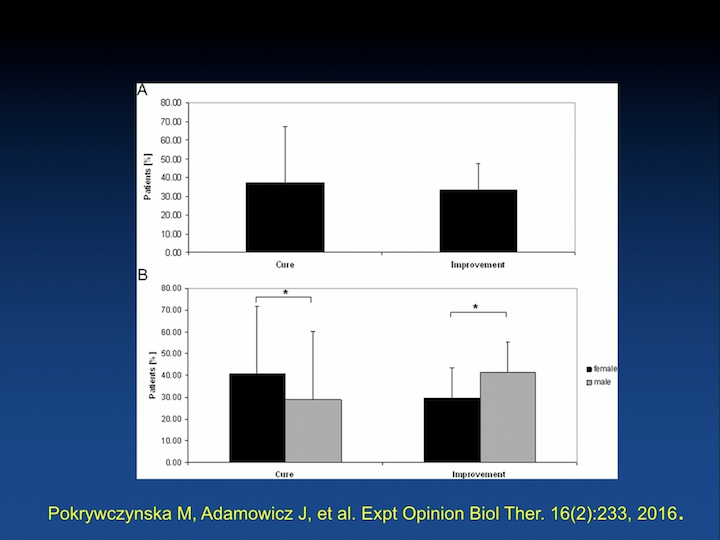
Metanalysis n=682
Since then, there have been a number of studies. This is a meta analysis published by a Polish group that looked at, there’s about 682 human patients that have been injected with some sort of stem cell.
Metanalysis Results
And, the results are pretty much similar, about a 40% improvement, 50%, 40%, 30% cure rate, very few studies that have looked at long-term outcomes.
15 Trials
There’s about 15 clinical trials in ClinicalTrials.Gov using stem cells for the treatment of incontinence.
The Journal of Urology
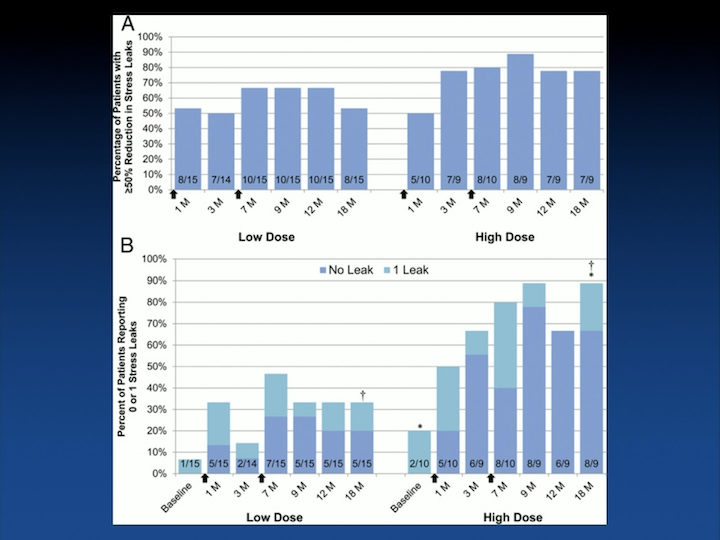
The most important trials for the U.S. market, or for the U.S., is going to be– are the ones affiliated with Mike Chancellor and – – myocyte. The original studies were really looking at safety and looking at different cell doses, and proved that the higher dose, the patients tended to do a little better.
Injections and Leakage
But that it was safe regardless. Again, the light blue in the lower graph shows one leak. As you can see, the more number of cells that you inject, the more likelihood that you have less leakage.
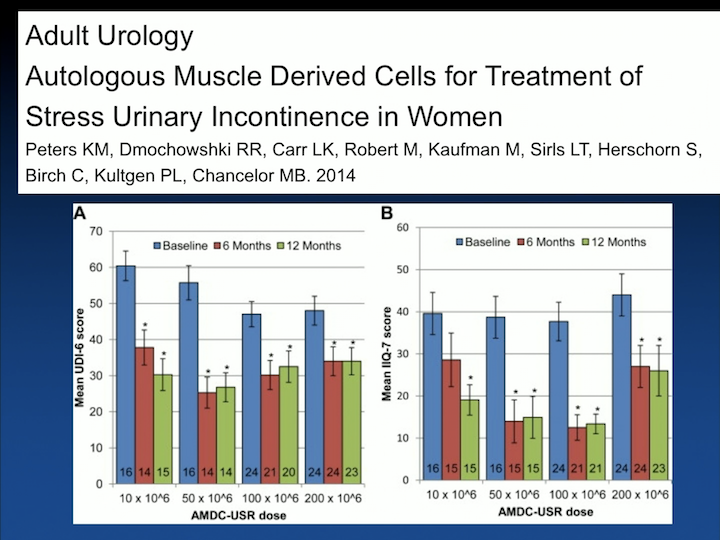
Adult Urology
The second study that published from the same group was a randomized clinical trial, randomizing patients with incontinence, so not in treatment arms but just to different doses of cells again to look at safety and look– trying to optimize what is the number of cells you will need to inject to be feasible.
Autologous muscle derived cell for urinary sphincter repair for recurrent or persistent SUI
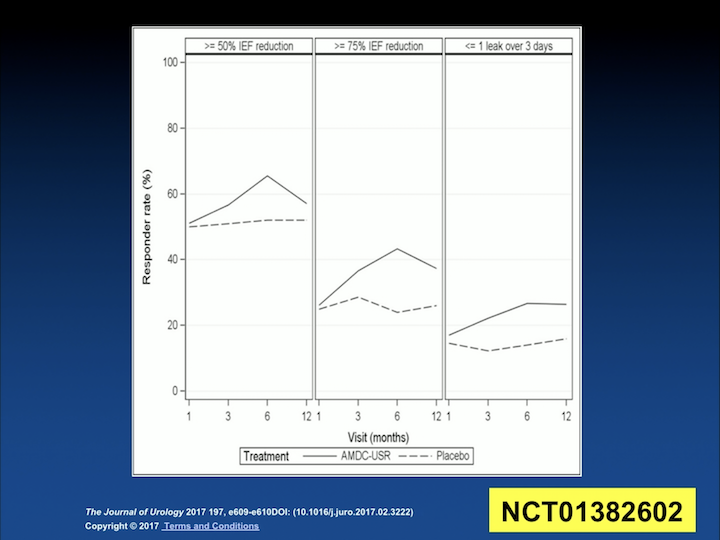
The last study that I’m aware of was presented at the AUA last year. Having seen the publication, I actually searched for it yesterday, and I couldn’t find it. But it must be in the works, but it was presented at the AUA last year, and it was an abstract where they looked at 143 women that were randomized to treatment, or no treatment, or kind of a vehicle placebo injection. So, they settled to the 150 times 10 to the 6th cells, and they– a small group of patients also were randomized, receiving one treatment or a repeat treatment at six months. Unfortunately, the study was ended early because the placebo effect was huge. And this is some of the data that they have.
AUA Study Outcomes
Originally, in the left column, this is how they defined pre-study, their outcomes, and as you can see, the placebo is the interrupted line, while the treatment is the solid line. And, they were so close to each other that they have tried to play with the definition of improvement to try to separate those two groups. But, as you try to separate them, you also decrease who is a responder and who is not a responder. So it is unclear to me, but this is really going to inform them how to proceed with their Phase 3/Phase 4 clinical trials. But I, again, I haven’t seen the details of the publication.
Summary
So in summary, these are injectable therapies, which are a mixed population of cells. There’s a number of animal studies that show improvement in urethra resistance. The clinical trials are promising. So, if you ask me, have we made progress in the past decade and a half, absolutely.
Which Cells?
We know that we’re not going to use embryonic stem cells. We can’t justify it for the treatment of stress incontinence. Almost no research has been done in induced stem cells, so it’s most likely going to be in adult stem cells that have really been shown to be safe in the short-term.
Which patient?
And when we look at the patient that we want to treat, looking at anatomic that lacks support, skeletal rhabdosphincter, and all the areas that we mentioned before, when we look at really patient driven therapy, is this going to help patients that have muscle problems, nerve injury, vascular injury? Really, the anatomic or the central nervous system is likely not going to be improved with this therapy. So, I am going to because of the sake of time, I can see my timer there, I cannot – I’m not going to be able to talk to you a lot about the fascia. It’s mostly to replace mesh. There’s a lot of studies that have been done now, not so much with stem cells, with the materials to try to replace mesh after a mesh debacle that we had in our field.
Mechanism of Action
And, what I do want to say is that we really do not understand how these cells work, how these therapies work on any mean. And, if we’re looking at mechanism of action, we’re really not ready for this therapy in terms of understanding it. But, just because we don’t understand how it works doesn’t mean it doesn’t work.
JAMA/THE LANCET
It’s being used in a number of conditions. In diabetes it’s being used. In macular degeneration.