John W. Davis, MD, presented “Focal Therapy for Localized Prostate Cancer: Is There a “Middle Ground” Between Active Surveillance and Definitive Treatment? ” during the 29th Annual International Prostate Cancer Update on January 26, 2019 in Beaver Creek, Colorado.
How to cite: Davis, John W. “Focal Therapy for Localized Prostate Cancer: Is There a “Middle Ground” Between Active Surveillance and Definitive Treatment?” January 26, 2019. Accessed Apr 2024. https://grandroundsinurology.com/using-high-resolution-micro-ultrasound-for-prostate-biopsies-for-active-surveillance/
Focal Therapy for Localized Prostate Cancer: Is There a “Middle Ground” Between Active Surveillance and Definitive Treatment?
John W. Davis, MD, discusses the lack of consensus regarding the definitions and benefits of focal therapy for prostate cancer. He defines the patients who would benefit most from focal therapy, as well as overcoming current challenges in this approach.
Abstract:
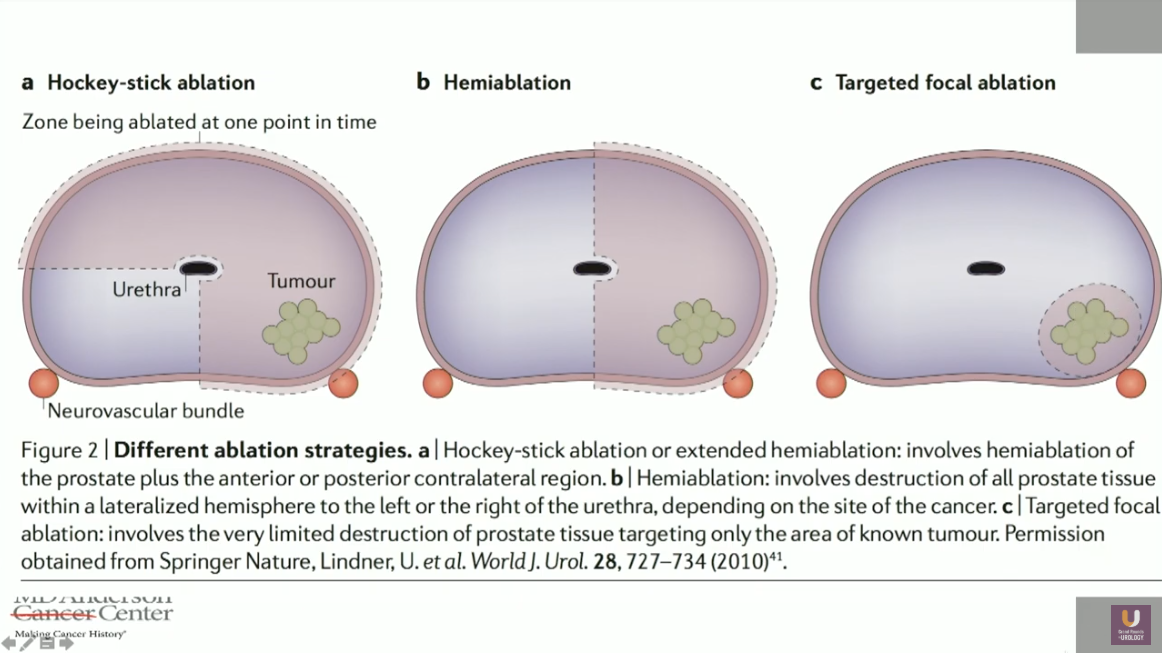
“Targeted focal destruction of cancer tissue with preservation of surrounding healthy parenchyma” is a definition proposed by Perera et al. for focal therapy. This definition currently overlaps with three different concepts: lesion ablation with a margin, hemiablation, and hemiablation of the prostate plus the anterior contralateral region, or in other words, “hockey stick” ablation.
It is critical to refine these concepts, gather more data on ablation technologies, and uniformly define endpoints of focal therapy.
Likely, focal therapy has a minimal benefit in cancer survival. However, it may be worthwhile, especially in Gleason 3+4 disease, as focal therapy may prevent disease upgrading.
One major dilemma in focal therapy stems from the fact prostate cancer is mostly a multifocal disease. The main method for guiding focal therapy, MRI, currently provides more estimation than precision. A proposed compromise is to perform deep saturation biopsies to select candidates and proceed with either a lesion directed focal therapy with a generous margin, or perhaps hemiablation.
Presently, evidence shows little gain from treating low-risk disease with focal therapy as opposed to definitive therapy or active surveillance. Furthermore, possible oncologic compromise could result from attempting focal therapy in unfavorable-risk/high-risk disease.
About the International Prostate Cancer Update
The International Prostate Cancer Update (IPCU) is an annual, multi-day CME conference focused on prostate cancer treatment updates. The conference’s faculty consists of international experts, and the event caters to urologists, medical oncologists, radiation oncologists, and other healthcare professionals. Topics encompass prostate cancer management, from diagnosis to treating advanced and metastatic disease. Dr. Davis presented this lecture during the 29th IPCU in 2019. Please visit this page in order to learn more about future IPCU meetings.