Thomas J. Pugh, MD, presented “New Considerations for Oligometastatic Prostate Cancer” at the International Prostate Cancer Update on January 27, 2018 in Beaver Creek, Colorado
How to cite: Pugh, Thomas J. “New Considerations for Oligometastatic Prostate Cancer” January 27, 2018. Accessed. https://grandroundsinurology.com/New-Considerations-for-Oligometastatic-Prostate-Cancer/
Summary:
Thomas J. Pugh, MD, summarizes the evolution oligometastatic prostate cancer management, the current definition of the oligometastatic state, and clinical trials for using stereotactic radiation therapy (SBRT) in this setting.
Reveal the Answer to Audience Response Question #1
Which of the following is the best current definition of oligometastatic prostate cancer?
- A. ≤ 3 lesions outside of the pelvis
- B. ≤ 5 lesions limited to lymph nodes or bones
- C. ≤ 10 total lesions without visceral organ involvement
- D. The definition is evolving and may be dependent upon imaging modality
Reveal the Answer to Audience Response Question #2
Which of the following is true regarding SABR metastases directed therapy for M1 prostate cancer?
- A. Retrospective studies suggest local control of the targeted lesion is above 90%
- B. Retrospective studies suggest Grade 2+ toxicity is <10%
- C. SABR significantly improves prostate cancer specific survival compared to best systemic therapy alone
- D. All of the Above
- E. A and B only
(Twitter Question) Reveal the Answer to Audience Response Question #3
True or False- Prospective randomized trials have demonstrated superior overall survival following radical therapy to the prostate in M1 prostate cancer.
- A. True
- B. False
New Considerations for Oligometastatic Prostate Cancer – Transcript
Click on slide to expand
Case
Okay, so this is the case we’ll focus on. It’s a 59-year-old man otherwise healthy. He presented with lower urinary tract symptoms, PSA 23, on DRE. He had a T3 examination and he had TRUS guided biopsy that showed Gleason grade 4+5 prostate adenocarcinoma 12 out of 12 cores.
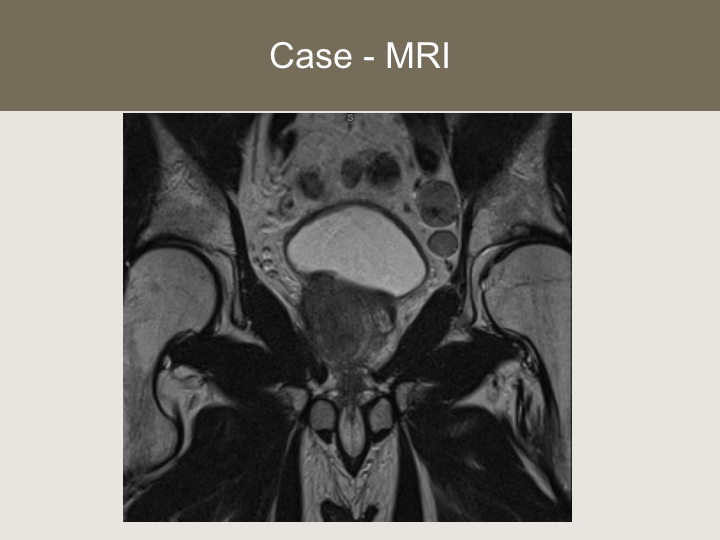
Case – MRI
Here is his MRI. This is a coronal projection. As you can tell, he’s got clear extracapsular extension. He’s got some enlarged pelvic lymph nodes all of which were below the aortic bifurcation.
Tc-99 Bone Scan
He had a whole body Tc-99 bone scan that showed radiotracer uptake in the right 7th rib. This was sclerotic on CT. We did the CT-guided biopsy of that lesion, and it turned out that it was positive for prostate adenocarcinoma. We’re going to skip those. We did those already.
If the horse is out of the barn
So even just a few years ago in this situation we used to throw our hands in the air and say this is metastatic cancer, it’s incurable, we’re going to give you some medicine. It will keep the cancer at bay for a while. Eventually the medicine won’t work. We’ll switch to a different medicine, and eventually you’re going to die of prostate cancer. I think that dynamic has radically changed and thus the concept of management of oligometastatic disease.
3-Tiered Approach to Curative Intent Therapy for M1 Disease
In order for a curative approach to be successful, there really is a three-tiered approach that is necessary in those who have M1 disease. The cornerstone of that is effective systemic chemo hormonal therapy, which we’re not going to talk a whole lot about during my presentation because we’re going to talk a lot about that the rest of the morning. But that clearly is the cornerstone of treatment where we’re affecting the cancer cells we can’t see on advanced imaging.
Number two is some form of metastases directed ablative therapy, either cryoablation, HIFU, I think the most mature of which is stereotactic body radiation therapy or SABR.
And then three is some sort of extirpation or sterilizing local therapy to the primary site.
What is “oligometastatic” and is the term meaningful?
So oligometastatic disease, we touched on this a little bit, but the traditional definitions are based upon the numbers of lesions on Tc-99 bone scan, and occasionally we reference anatomic site of involvement. There does appear to be a good amount of biological variability, and there are some molecular studies that have looked at the different genomics of the metastasis versus the primary tumor, and so just because we see images that suggest oligometastatic disease, the biological behavior can actually be quite different. More recently, novel imaging tools have come to fruition, including whole body MRI, novel PET radiotracers that really change the way we think about oligometastatic disease. An example of that was in the journal of nuclear medicine just last year by the Cornell group where they scanned the patient with Tc-99 bone scan that we’re all used to. That’s the image on your left and then used a ligand PSMA-based radiotracer image and in the same patient we have two dramatically different images where we’re finding osseous metastasis, visceral metastasis, and so I think this is a moving target.
Select Contemporary Studies Defining Oligometastatic PCA
There are a few studies out there that help us define the oligometastatic state. The first several of these are studies that have been published that primarily looked at lesions that were less than 3 to 5 in number and primarily bone only. They either use sodium fluoride PET or Tc bone scan and then I added a few ongoing clinical trials, a few tertiary cancer centers, and you can see the imaging modalities aren’t necessarily specified, and the number of lesions and where those lesions are located are quite variable.
Which of the following is the best current definition of oligo-metastatic prostate cancer?
So I think the answer to this question is that D the definition is evolving, and I don’t quite know what oligometastatic prostate cancer is right now.
Metastases Directed Therapy
Okay. Let’s move on to how to treat these patients and our sample patient. We’ll start with metastases-directed therapy.
Rationale for Metastases Directed Therapy
So the rationale for doing something like this, delivering aggressive therapy to metastatic disease is, one, early ablation of metastatic disease may reduce the risk of skeletal events or other complications from disease progression. Metastatic lesions, again they have different molecular phenotype, and that may translate to systemic therapy resistance and by targeting these lesions, we may delay or defer castrate resistance, and perhaps we delay the initiation of cytotoxic chemotherapy or other therapeutic augmentation. Of course, the goal of this, the end game is to improve oncologic outcome under the very simplistic principal that less cancer in the body is a good thing, because few of us have seen patients die of prostate cancer with a low disease burden.
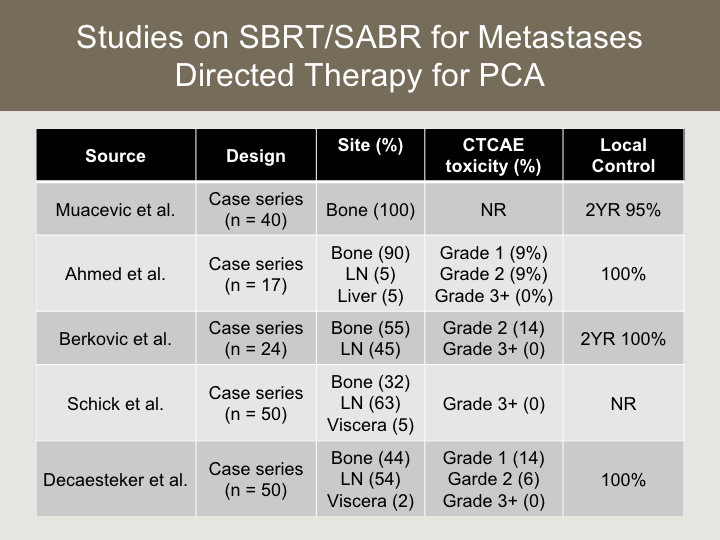
Studies on SBRT/SABR for Metastases Directed Therapy for PCA
In terms of studies on SBRT, which is probably the most mature of which, there’s other modalities that we can use to ablate metastatic lesions. There’s several on using SBRT, which is essentially radiosurgery for metastatic lesions. There’s five that are published. These are all case series. So they’re all retrospective designs. The numbers are still relatively small. They’re predominantly bone lesions that were targeted, but it includes some visceral lesions, and several that involved lymph nodes as well. And what I want you to take from this slide is that the evidence to date would suggest that this is a safe thing to do, where we really haven’t seen any grade 3 or higher toxicity, and the risk of grade two or higher toxicity is quite low, and in the studies that did report local control, specific to the targeted lesion we see excellent ability to sterilize whatever we’re targeting with this type of intervention.
So overall, this is a very well-tolerated treatment that seems to control the lesion quite well. We don’t have great evidence as to how this affects harder oncologic outcomes. We do know that in two of these studies where they did look at freedom from androgen deprivation, where they actually took some of these patients off of hormone therapy and allowed for testosterone recovery that it was around 2 to 3 years freedom from androgen deprivation in those patients that were treated with this aggressive strategy. These are not comparative studies, so it’s hard to place too much stock in that.
Summary: Metastases Directed Therapy
So in summary of metastasis-directed therapy, ablative metastasis-directed therapy such as SBRT are associated with minimal toxicity risk, and they provide excellent local control. The current data are insufficient for determining the effect of such strategies on oncologic outcomes.
Which of the following is true regarding SABR/SBRT metastases directed therapy for M1 prostate cancer?
And so most of you I think got this right where we just don’t know how these interventions affect cancer outcomes yet, but they do appear to be safe, and they do appear to offer excellent local control.
Management of the Prostate
This is a really sexy topic right now in terms of managing the prostate in metastatic disease. There’s a lot of work being done to facilitate prospective trials. The rationale for treating the prostate is improved pelvic disease control or prophylactic palliation. I think we’ve all seen patients who have uncontrolled pelvic disease, and it’s exquisitely morbid. So this is a methodology of getting in front of that. A very hot topic right now is how treating the primary tumor might augment host immunosuppression and potential anti-cancer immunomodulation. This field of immunotherapy is very interesting space, and then of course what we are looking to do, similar to what we see when we do cytoreductive nephrectomies in renal cell carcinoma is by perhaps treating the mother ship we may improve oncologic outcome.
NCDB Analysis
There is no prospective studies on this topic. There is a good amount of retrospective evidence. We undertook a project at University of Colorado and incorporated a multi-institutional group where we used the National Cancer Database to conduct a query where we looked at men who were diagnosed with M1 prostate cancer all of whom received up front ADT. We excluded cryotherapy and brachytherapy as a local intervention, and we excluded patients who died within one month of diagnosis. And we used standard statistical tools including propensity score matching and landmark analysis to determine what factors were associated with improved outcome.
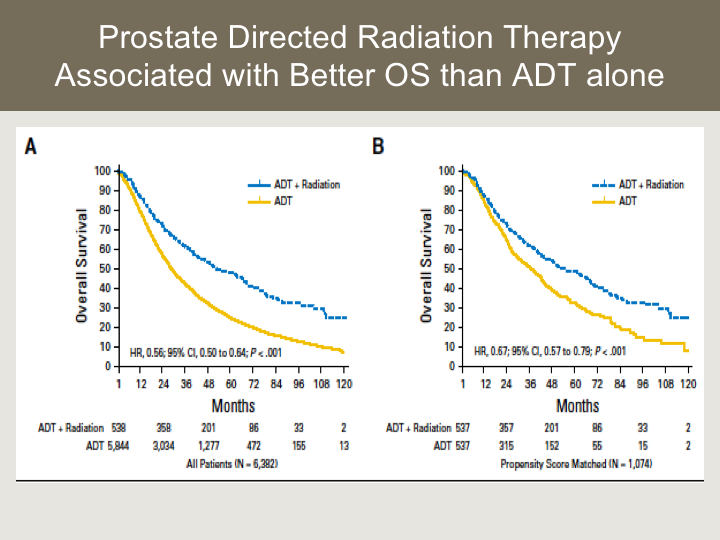
Prostate Directed Radiation Therapy Associated with Better OS than ADT alone
Here are the results of that analysis, so we were able to identify over 3,000 patients through the database. There is good radiation detail through this database. That’s one of the benefits of it, and you can see in Figure 8, there was an overall survival advantage attributable to radiation therapy but compared to hormone therapy alone that was fairly dramatic reducing the risk by half, and when we did propensity score matching that difference persisted and was approximately a third.
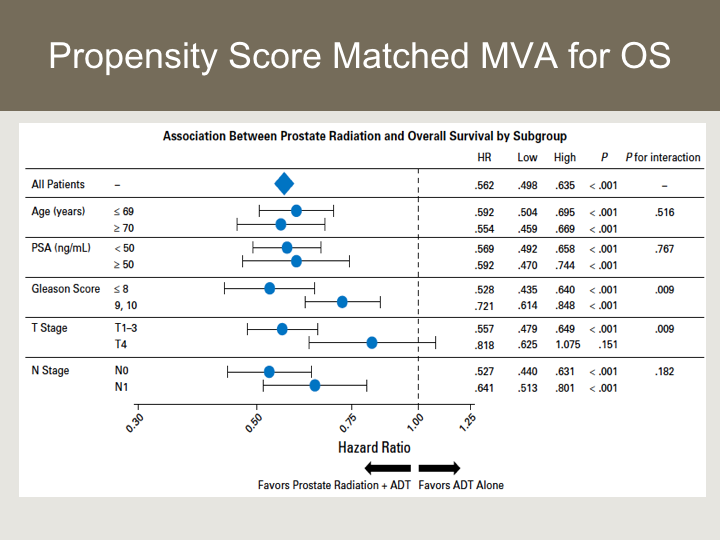
Propensity Score Matched MVA for OS
This is the Forest plot looking at associations with radiation versus hormone therapy alone, and you can see that regardless of subset with the exception of patients who had a T4 lesion, again the numbers were relatively small, all of these subsets seemed to benefit from the local therapy.
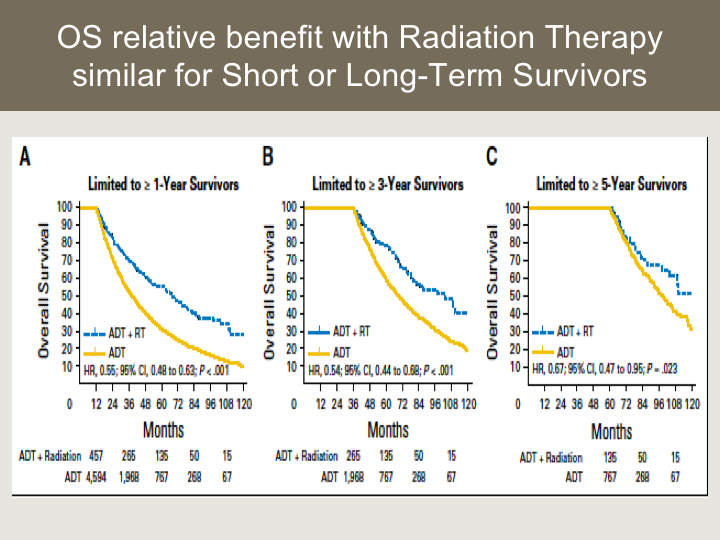
OS relative benefit with Radiation Therapy similar for Short or Long-term Survivors
This is the landmark analysis where we tried to control for selection bias and perhaps the patients who were being selected for local therapy had better prognosis and longevity, and so what we found was that the benefit appeared to be similar for long-term survivors versus shorter-term survivors.
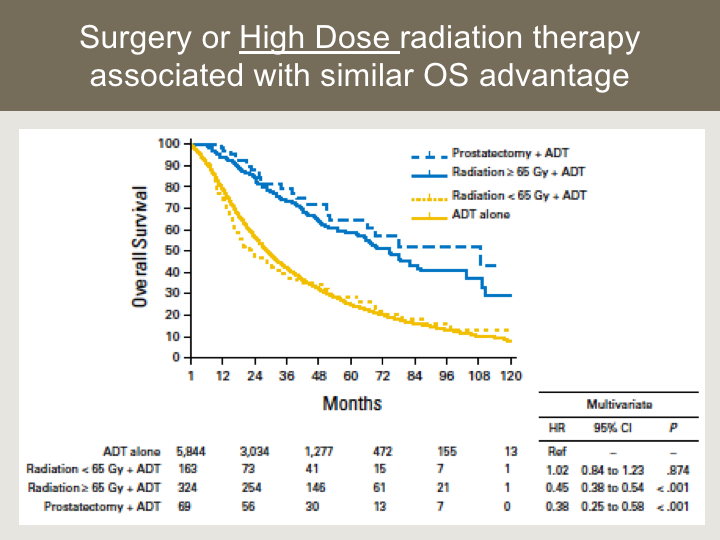
Surgery or High Dose Radiation Therapy Associated with Similar OS Advantage
We used a prostatectomy group as well from this data set as a comparative. This is a fairly interesting result so what we found is that in the prostatectomy and hormone therapy group they had comparable, not significantly different survival outcomes compared to those who received high-dose radiation defined as above 65Gy, 65Gy or more, whereas if the patient received less than 65 Gy, more consistent with a palliative dose of radiation, the outcomes for survival were similar to hormone therapy alone. So, this suggests that ablative therapeutic interventions to the prostate or the primary tumor are what really confers the the potential advantage in this space.
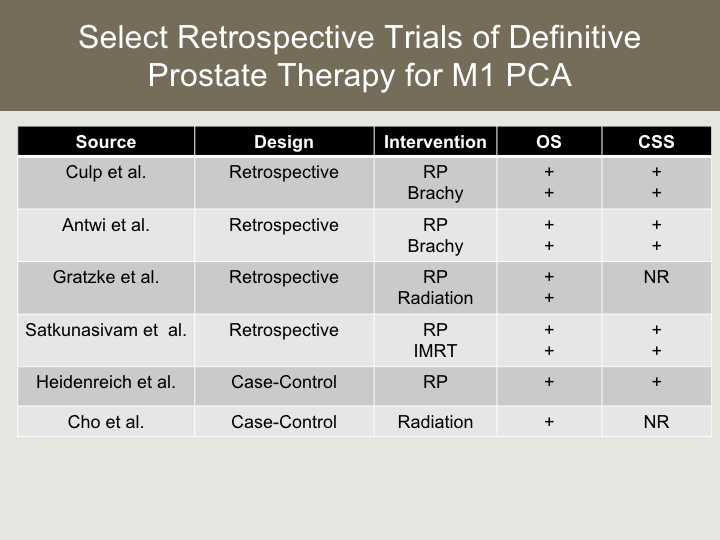
Select Retrospective Trials for Definitive Prostate Therapy for M1 PCA
We’re not the only group to find this. There’s actually a plethora of retrospective studies to show the same thing. These are very large population datasets that looked at radical prostatectomy, brachytherapy, external radiation IMRT, all of which showed a similar result in terms of better overall survival in the group that received therapy to the primary, and some of those studies attributed that improved overall survival to disease or cause specific survival improvements, and there are two case-control series that looked at prostatectomy and radiation respectively, also showing similar results.
Summary: Prostate Directed Therapy for M1 PCA
So in terms of prostate directed therapy for M1 prostate cancer, radical treatments to the prostate have a well-defined safety profile, and they likely reduce the risk of pelvic morbidity and need for subsequent palliative interventions.
Primary debulking might augment the communicating ecosystem of metastatic prostate cancer. Additional work needs to be done in this space, and there are several retrospective studies to support oncologic outcome improvement in men receiving radical therapy to the primary, but I’d like to emphasize that these results really need to be applied cautiously because of the retrospective design and the potential for selection bias, and the likelihood that selection bias and other non-controlled factors influenced the results.
Prospective randomized trials have demonstrated superior overall survival following radical therapy to the prostate in M1 prostate cancer:
I think the answer to this question currently of our randomized answer is that we don’t have prospective data in this space yet. We just have a fairly compelling body of retrospective evidence that I think supports having these conversations with patients, but we do need to do the randomized studies to demonstrate that this benefit truly exists. Thanks for your attention and the people in the back for calling the audible.
ABOUT THE AUTHOR
Dr. Pugh is an Associate Professor of Radiation Oncology and Vice Chair of Clinical Affairs at the University of Colorado Anschutz Medical Campus in Denver. He is a board-certified radiation oncologist specializing in cancers of the genitourinary tract, including malignancies of the prostate, urinary bladder, testicle, and kidney. Dr. Pugh moved to the University of Colorado Cancer Center from the MD Anderson Cancer Center in Houston, where he had been a faculty member for over five years.
Dr. Pugh obtained his undergraduate degree from the University of Kansas, which he attended on a basketball scholarship. He was one of seven athletes nationally to be awarded a post-graduate scholarship by the national collegiate athletic association (NCAA) for academic and athletic achievement. He obtained his medical degree from the University of Colorado and spent a year as an internal medicine intern at the University of Colorado Hospital. He completed his residency training in Radiation Oncology at the University of Colorado Hospital, where he served as Chief Resident. He completed a prostate brachytherapy fellowship at the Seattle Prostate Institute in Seattle, Washington, sponsored by the American Brachytherapy Society. Dr. Pugh’s interests include advanced technology integration into cancer therapeutics, prostate brachytherapy, particle therapy including proton beam therapy, image-guided radiation therapy (IGRT), stereotactic body radiotherapy (SBRT), intensity modulated radiotherapy (IMRT), and integration of medical therapeutics, including chemotherapy and hormone therapy, with radiation therapy. His research has focused on improving quality of life and reducing side effects for men with prostate cancer. In addition to his interest in technical advances in radiation oncology, he is involved in the development of cancer treatment clinical trials for patients with genitourinary cancers.
Dr. Pugh is an internationally recognized expert in genitourinary cancer and has served in numerous leadership roles on multiple ASTRO, ABS, and RTOG/NRG committees. He has served as principal investigator on several institutional and national clinical trials, including MDACC 2012-1003, MDACC 2012-1013, RTOG 0815, RTOG 1115, and MDACC 2013-0555. He has published over 100 peer-reviewed articles, abstracts, and book chapters.