M. Scott Lucia, MD, presented “Mapping Pathology as a Guide to Focal Therapy: The Role of Mapping Biopsies and Tissue Markers” during the 23rd Annual Southwest Prostate Cancer Update on April 15, 2018 in Scottsdale, Arizona
How to cite: Lucia, M. Scott. “Mapping Pathology as a Guide to Focal Therapy: The Role of Mapping Biopsies and Tissue Markers” April 15, 2018. Accessed [date today]. https://grandroundsinurology.com/the-role-of-mapping-biopsies-and-tissue-markers/
Mapping Pathology as a Guide to Focal Therapy: The Role of Mapping Biopsies and Tissue Markers – Summary:
M. Scott Lucia, MD, discusses patient selection for targeted focal therapy (TFT) for prostate cancer, methods for accurately assessing tumors, as well as a pathological approach to monitoring the efficacy of TFT. Specifically, he reviews the accuracy of transperineal template-guided mapping biopsies (TTMB), Multiparametric MRI (mpMRI), and biomarkers.
Identifying Candidates for Prostate Targeted Focal Therapy
While there is no consensus on the ideal candidate for TFT, Dr. Lucia’s proposes a spectrum model for identify level of patient eligibility for this method. Pathology features, such as grade of disease, tumor volume, and unilateral location, determine where a patient fits on the spectrum.
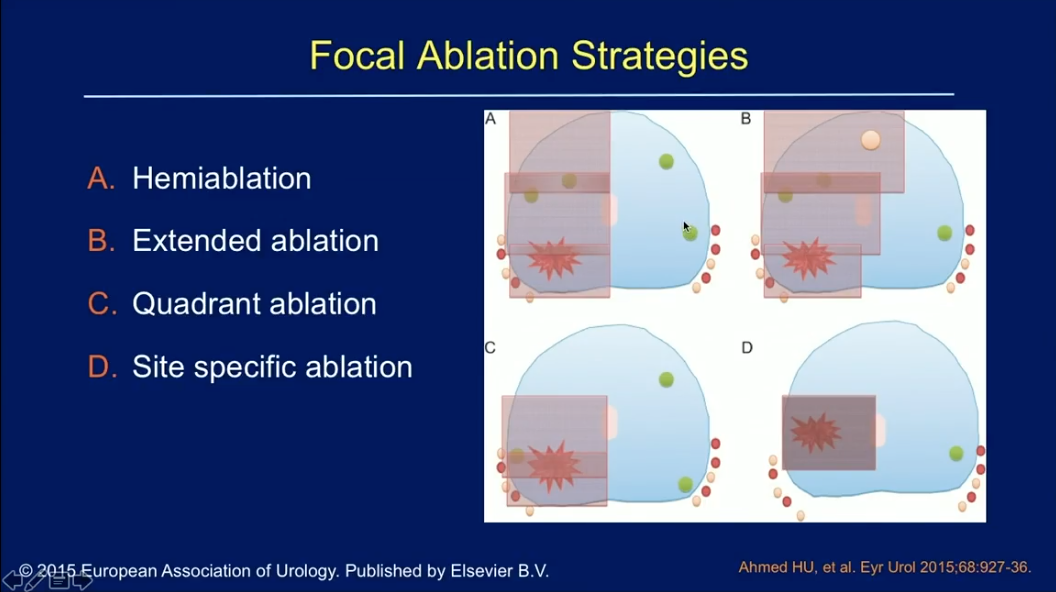
Focal Ablation Strategies and the Limitations of Transrectal Ultrasound (TRUS)
There are four major prostate focal ablation strategies: hemiablation, extended ablation, quadrant ablation, and site specific ablation. As the concept of TFT dictates, all these strategies may leave behind low-grade, insignificant cancer. Physicians must be certain that these untreated cancers are truly insignificant in order to responsibly employ this method. Unfortunately, current modalities for determining tumor aggressiveness, extent, and precise location are insufficient.
For example, TRUS biopsies often leads to false negatives and cancer upgrading. Cancer sampling is a function of the tumor volume to prostate volume ratio. Similarly, high-grade tumor sampling is a function of the high-grade component to prostate volume ratio. TRUS has a higher likelihood of finding a significant cancer in small prostates with large tumors and large high-grade components. Conversely, patients with large prostates and smaller volume of disease are more likely to have false negatives on TRUS. Additionally, TRUS relatively undersamples the anterior prostate.
Because of these limitations, TRUS may miss the index lesion and the highest-grade cancer tissue in the prostate, leading to understaging and undergrading.
Solutions to Missed Insignificant Cancers in Biopsy and Treatment
Transperineal Template-guided Mapping Biopsies (TTMB)
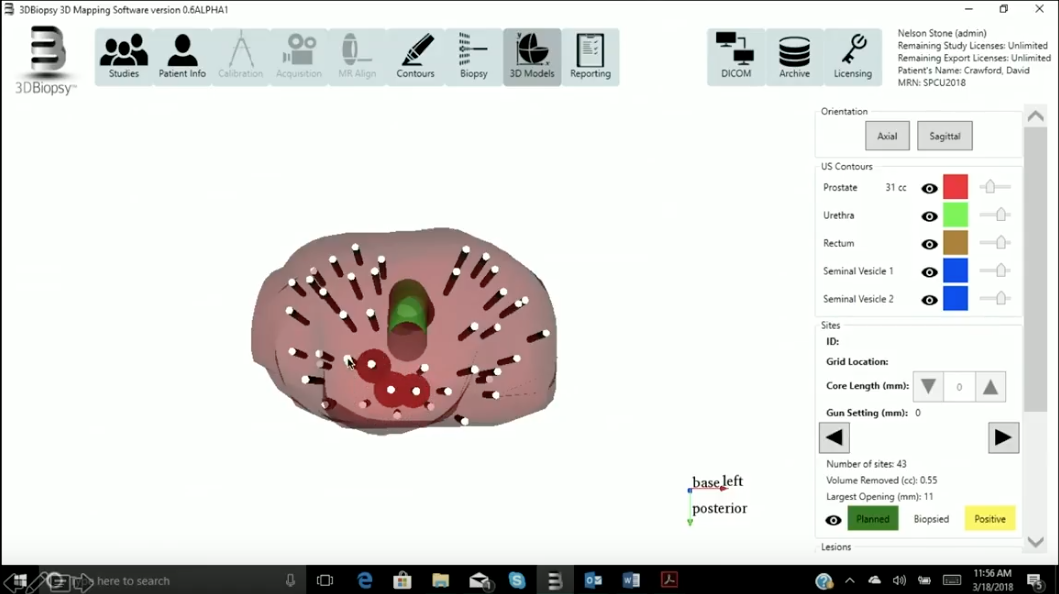
To address the need of accurately assessing tumor grade, aggressiveness, extent, and location throughout the prostate before continuing to TFT, Dr. Lucia suggests TTMB. This method involves using a brachytherapy template in the transperineal region. Physicians take cores from every point on a 5 mm spaced grid in order to uniformly sample the entire prostate. He then describes a method of sending taken cores to pathology by differentially inking them and fitting them into a single cassette. This significantly lowers financial costs. Using pathology reports and 3D mapping software, physicians can then draw the area of a lesion in the gland to target.
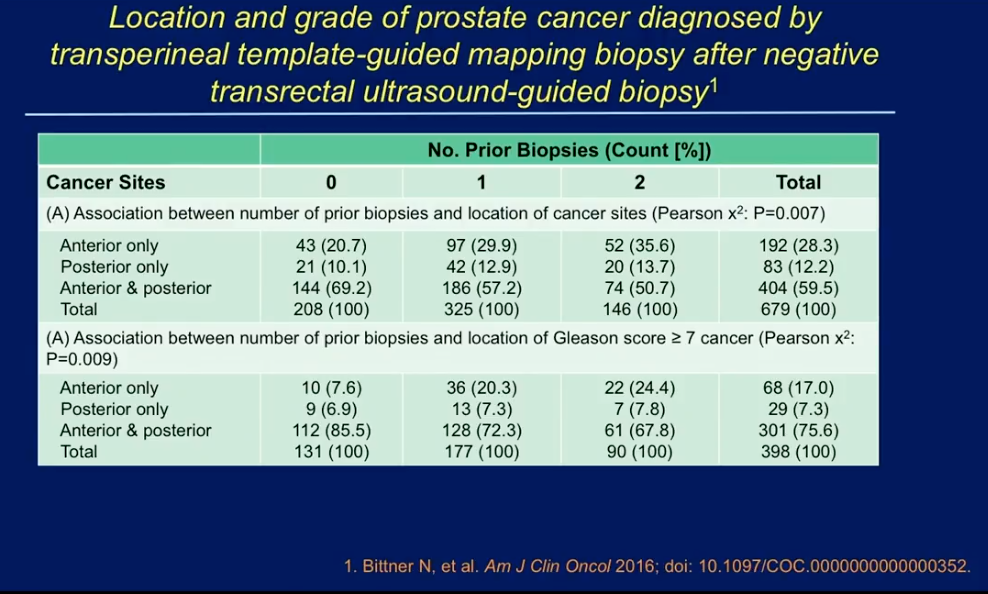
A study by Valerio M. et al. compared the risk stratification of patients diagnosed with prostate cancer by TRUS biopsy versus their subsequent TTMB. The results of show a restratification of patients into high- and intermediate-risk categories after TTMB in those who had more low-risk disease on initial TRUS biopsy. Similarly, a multi-institutional study by Bittner N. et al., found 679 patients with positive TTMB despite having a negative TRUS biopsy. These data suggest TTMB finds more significant cancer than TRUS biopsy will show on the same patient
Multiparametric MRI (mpMRI)
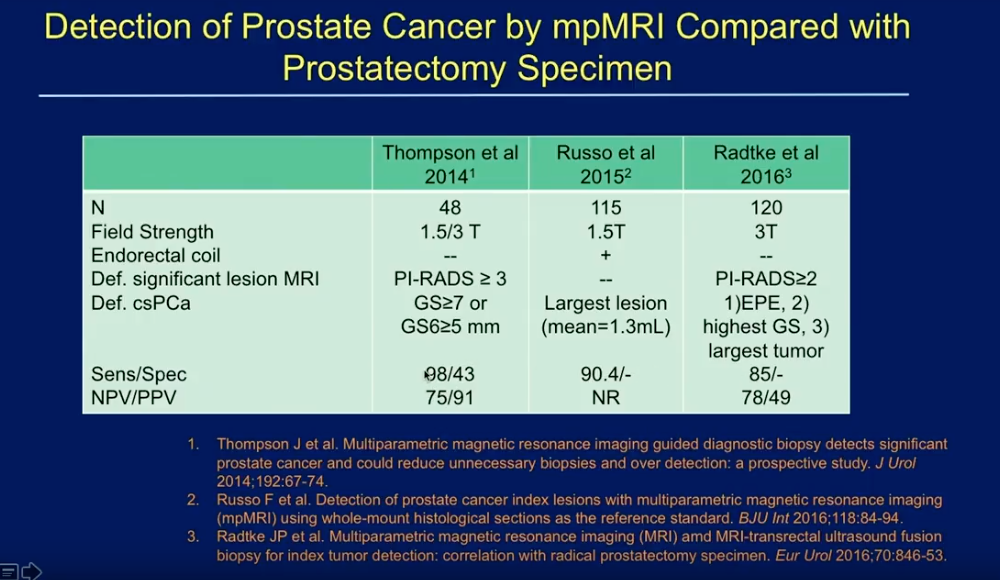
Dr. Lucia three studies observing mpMRI as a tool to improve the delivery of TFT, as pictured on the left. These studies show sensitivity rates for index lesions of mpMRI ranging from 85-98, and negative predictive values ranging from 75 to 78. However, these studies had limitations in assessing mpMRI due to inconsistency in field strength, use of endorectal coils, and definition of significant disease.
A study by Mortezavi et al. compared the ability of mpMRI to detect of prostate cancer versus TTMB with clinically significant disease defined as a Gleason score of 3+4 or above. Of 291 patients with a Likert score of 3 to 5, mpMRI guided biopsy detected cases of clinically significant prostate cancer in 129 patients, TTMB detected 176 cases, and a combined approach detected 187 cases. Overall, mpMRI alone would have missed 58 cases. Similarly, in a study by Sivaramen et al., which defined clinically significant disease as Gleason score 7 or above, TTMB identified tumors in 27 out of 74 who had prior negative MRI-TRUS biopsies. Of these tumors, 70.4% were significant lesions.
The Role of Pathology in Monitoring Efficacy of TFT
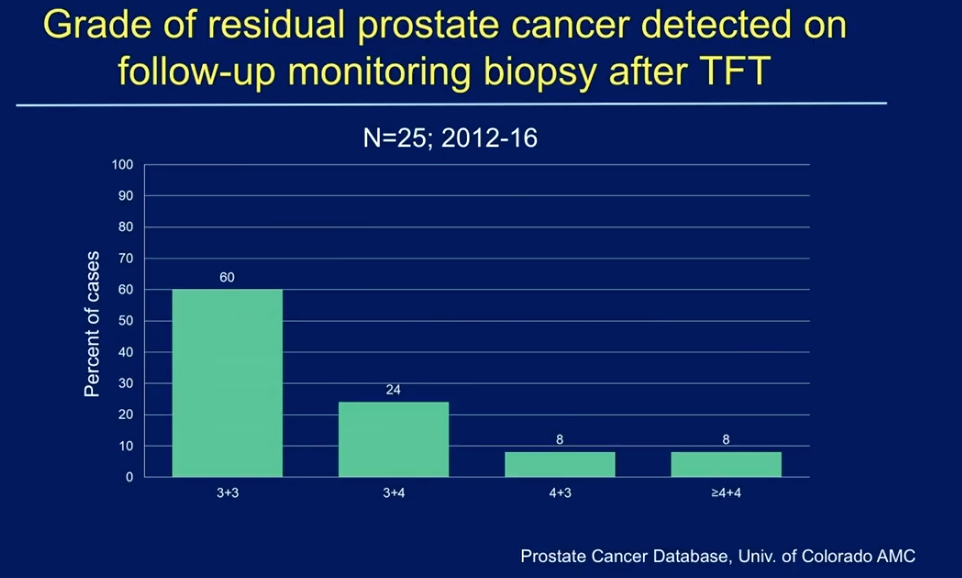
The University of Colorado, Dr. Lucia’s institution, monitors patients post-TFT with an active surveillance approach. Physicians follow patients’ PSA and taking an annual follow-up 12-core biopsy. The Prostate Cancer Database of the University of Colorado observed prostate cancer patients treated with TFT during 2012-2016. This data shows there have been 25 patients who have documented recurrence on follow-up biopsy. Out of those patients, 60% had recurrent disease with a Gleason score of 3+3. Unfortunately, some patients had Gleason score 4+3, 4+4, or even higher-grade cancers. It is unclear as to whether this was due to inadequate TFT, or if new cancers emerged since the initial therapy. However, the fact that 20% of the 25 patients with recurrences had 4-6 cores positive on follow-up biopsy suggests the emergence of new tumors.
Biomarkers like SelectMDx, 4KScore, and PHI testing may be useful for indicating which patients should have re-biopsy following the annual follow-up. If a re-biopsy does show recurrent disease, biomarkers such as the Prolaris and OncotypeDX can help evaluate if the patient should continue to salvage therapy.
Conclusions
Overall, pathological features are important for patient selection for TFT. While traditional transrectal schemes prove to be inaccurate, transperineal mapping biopsies offer improved pathological accuracy for grade and multifocality. Fusion biopsies and mpMRI may be useful for determining eligibility of focal therapy in some patients. However, those methods may underestimate tumor burden.
ABOUT THE AUTHOR
M. Scott Lucia, MD, is Professor and Vice Chair of the Department of Pathology and Director of Anatomic Pathology and of the Prostate Diagnostic Laboratory at the University of Colorado Anschutz Medical Campus (UCAMC) School of Medicine. He also serves as the Director of the Prostate Cancer Research Laboratories at UCAMC and of the UCAMC Biorepository Core Facility. Dr. Lucia received his MD from the University of Colorado School of Medicine in 1988. He completed his internship and residency in pathology at the University of Colorado in 1993. He was a research fellow in the Laboratory of Chemoprevention at the National Institutes of Health from 1993 to 1995, before returning to the University of Colorado in 1995.
Dr. Lucia served as the primary pathologist for the Prostate Cancer Prevention Trial (PCPT) and Vitamin E and Selenium Chemoprevention Trial (SELECT), sponsored by the Southwest Oncology Group; the Medical Therapy of Prostate Symptoms (MTOPS) trial, sponsored by the NIDDK; and the Reduction with Dutasteride of Clinical Progression Events in Expectant Management of Prostate Cancer (REDEEM), sponsored by GlaxoSmithKline. He directs the operation of several tissue and serum biorepositories for prostate and prostatic diseases, including those for the PCPT, MTOPS, SELECT, and the University of Colorado Cancer Center Prostate Biorepository. He has authored or co-authored over 180 peer-reviewed articles, reviews, editorials, and book chapters. His primary areas of interest include pathology of prostate cancer and hyperplasia, early detection and prevention of prostate cancer, prostate cancer biomarkers, and mechanisms of carcinogenesis.